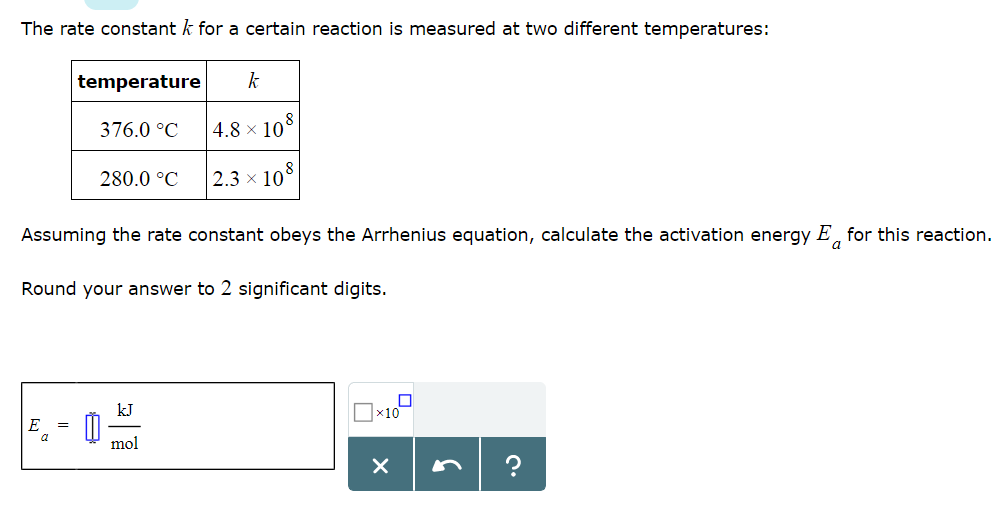
The Arrhenius equation’s use has also been extended to more complex biological systems, such as frog, beetle, and fly development, occasionally finding non-Arrhenius behavior (Krogh, 1914 Bliss, 1926 Bonnier, 1926 Ludwig, 1928 Powsner, 1935). The pre-exponential “frequency” factor A can be physically interpreted as proportional to the number of molecular collisions with favorable orientations. Based on this theory, the exponential term of the Arrhenius equation is proportional to the fraction of molecules with energy greater than the activation energy (E a) needed to overcome the reaction’s energetically unfavorable transition state. The Arrhenius equation would come to stand out from the rest, in part because it could be intuitively interpreted based on transition-state theory (Evans & Polanyi, 1935 Eyring, 1935a, 1935b Laidler & King, 1983). In the late 19 th century, scientists proposed many relationships between reaction rates and temperature (Berthelot, 1862 Schwab, 1883 Van’t Hoff & Hoff, 1884 Van’t Hoff, 1893 Harcourt & Esson, 1895). Thus, we find that complex embryonic development can be well approximated by the simple Arrhenius equation regardless of non-uniform developmental scaling and propose that the observed departure from this law likely results more from non-idealized individual steps rather than from the complexity of the system. In contrast, we find the two enzymes GAPDH and β-galactosidase show non-linearity in the Arrhenius plot similar to our observations of embryonic development. When we model multi-step reactions of idealized chemical networks, we are unable to generate comparable deviations from linearity. At low and high temperatures, however, we observed significant departures from idealized Arrhenius Law behavior. We find that the Arrhenius equation provides a good approximation for the temperature dependence of embryogenesis, even though individual developmental intervals scale differently with temperature. Here, we evaluate how well the simple Arrhenius equation predicts complex multi-step biological processes, using frog and fruit fly embryogenesis as two canonical models. Here is how the Arrhenius equation for backward equation calculation can be explained with given input values -> 0.01 = 10*exp(-(1.60217733000001E-17/(*0.5))).The famous Arrhenius equation is well suited to describing the temperature dependence of chemical reactions but has also been used for complicated biological processes. How to calculate Arrhenius equation for backward equation using this online calculator? To use this online calculator for Arrhenius equation for backward equation, enter Backward Pre-exponential factor (A b), Activation energy backward (E ab) & Absolute Temperature (T abs) and hit the calculate button. 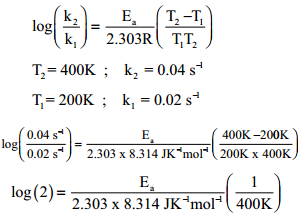 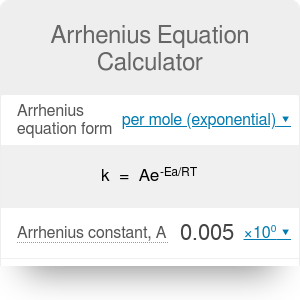
Backward reaction rate constant is denoted by K b symbol. How to Calculate Arrhenius equation for backward equation?Īrrhenius equation for backward equation calculator uses Backward reaction rate constant = Backward Pre-exponential factor* exp(-( Activation energy backward/( * Absolute Temperature))) to calculate the Backward reaction rate constant, The Arrhenius equation for backward equation represents the fraction of collisions that have enough energy to overcome the activation barrier (i.e., have energy greater than or equal to the activation energy Ea) at temperature T for a backward reaction.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
